State of RL for reasoning LLMs
Evolution of reinforcement learning for reasoning LLMs

I'm a PhD student working on (medical) multimodal learning, RL for LLMs, and everything I find interesting.
This website is where I share my thoughts on:
Feel free to connect with me on LinkedIn, Twitter or reach out via Email.
Evolution of reinforcement learning for reasoning LLMs
Showing that $0\neq 0$
Step-by-step exploration of nanochat's model
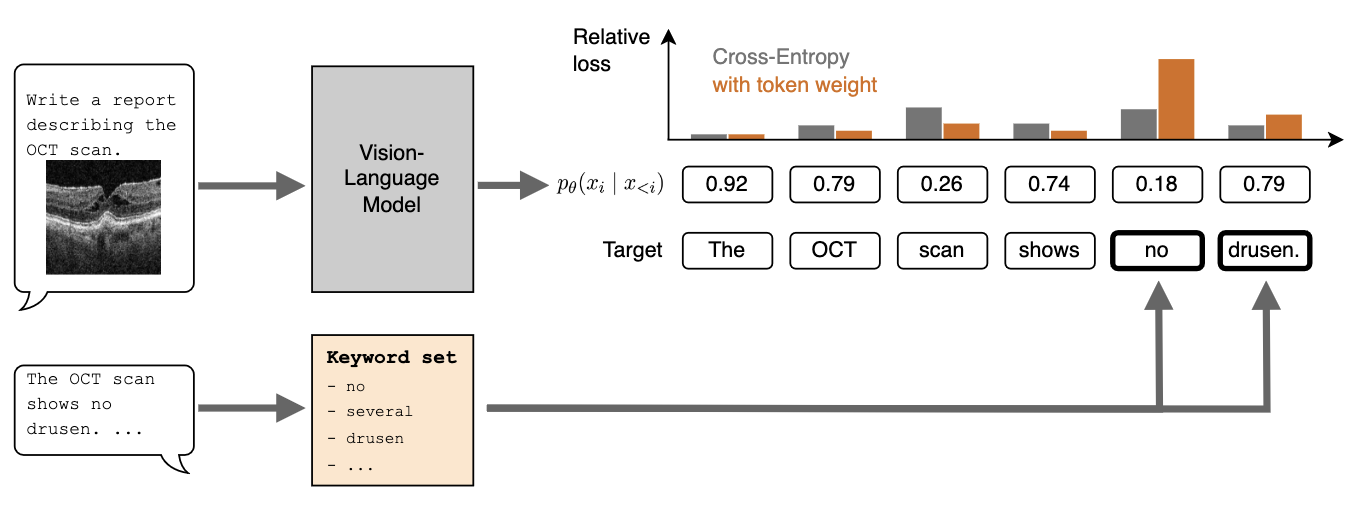
Medical Imaging with Deep Learning (Short Paper), 2026
Training vision-language models (VLMs) for medical report generation is often hindered by the scarcity of high-quality annotated data. This work evaluates the use of a weighted loss function to improve data efficiency. Compared to standard cross-entropy loss, which treats all token prediction errors equally, the reweighted loss shifts the focus to semantically salient tokens with outsized clinical importance. In experiments on ophthalmological report generation, we show that this simple method improves efficiency across multiple data scales, achieving similar report quality with up to ten times less training data.
Medical Imaging with Deep Learning, 2026
The histopathological analysis of whole-slide images (WSIs) is fundamental to cancer diagnosis but is a time-consuming and expert-driven process. While deep learning methods show promising results, dominant patch-based methods artificially fragment tissue, ignore biological boundaries, and produce black-box predictions. We overcome these limitations with a novel framework that transforms gigapixel WSIs into tissue-boundary aligned graph representations and is interpretable by design. Our approach builds graph nodes from tissue regions that respect natural structures, not arbitrary grids. We introduce an adaptive graph coarsening technique, guided by learned embeddings, to efficiently merge homogeneous regions while preserving diagnostically critical details in heterogeneous areas. Each node is enriched with a compact, interpretable feature set capturing clinically-motivated priors. A graph attention network then performs diagnosis on this compact representation. We demonstrate strong performance on cancer staging and survival prediction, outperforming methods with similar data requirements. Crucially, our data-efficient model (requiring less training data) achieves results competitive with a massive foundation model, while offering full interpretability through feature attribution.
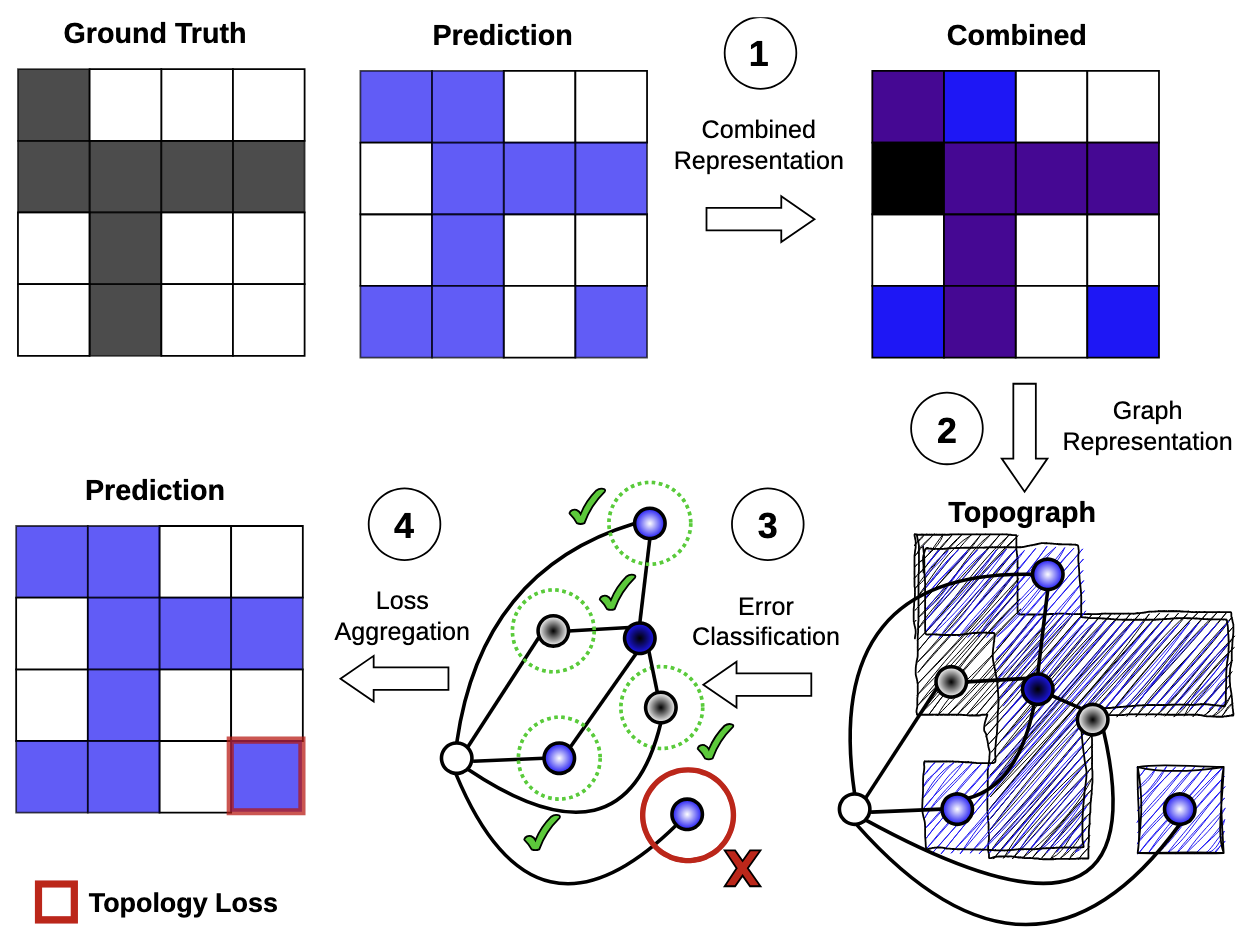
ICLR, 2025
Topological correctness plays a critical role in many image segmentation tasks, yet most networks are trained using pixel-wise loss functions, such as Dice, neglecting topological accuracy. Existing topology-aware methods often lack robust topological guarantees, are limited to specific use cases, or impose high computational costs. In this work, we propose a novel, graph-based framework for topologically accurate image segmentation that is both computationally efficient and generally applicable. Our method constructs a component graph that fully encodes the topological information of both the prediction and ground truth, allowing us to efficiently identify topologically critical regions and aggregate a loss based on local neighborhood information. Furthermore, we introduce a strict topological metric capturing the homotopy equivalence between the union and intersection of prediction-label pairs. We formally prove the topological guarantees of our approach and empirically validate its effectiveness on binary and multi-class datasets. Our loss demonstrates state-of-the-art performance with up to fivefold faster loss computation compared to persistent homology methods.
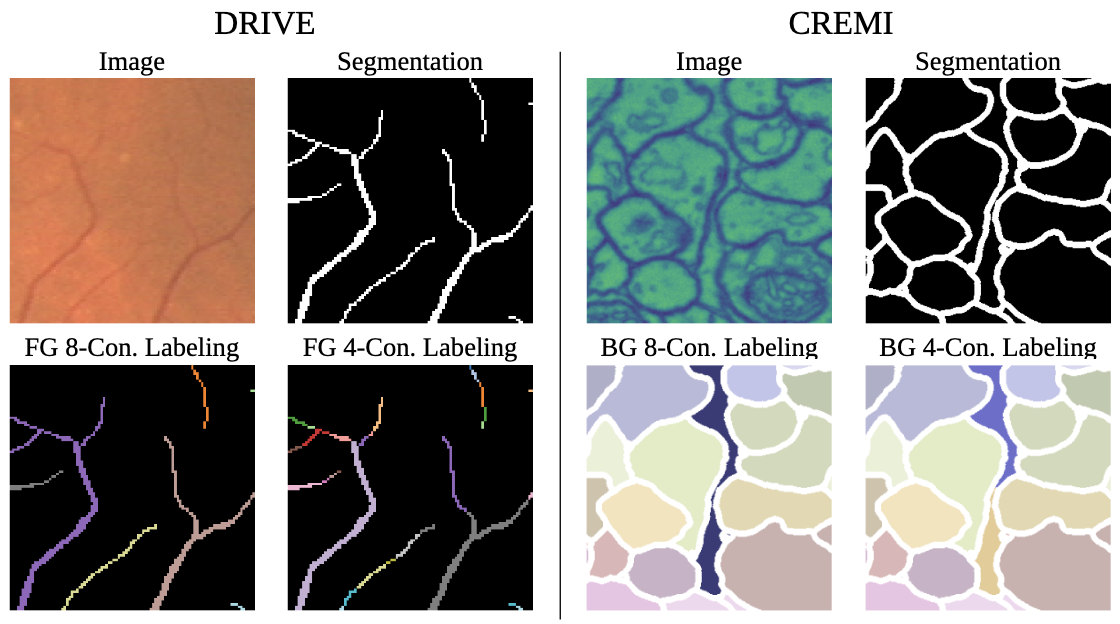
International Conference on Information Processing in Medical Imaging, 2025
Topological correctness, i.e., the preservation of structural integrity and specific characteristics of shape, is a fundamental requirement for medical imaging tasks, such as neuron or vessel segmentation. Despite the recent surge in topology-aware methods addressing this challenge, their real-world applicability is hindered by flawed benchmarking practices. In this paper, we identify critical pitfalls in model evaluation that include inadequate connectivity choices, overlooked topological artifacts in ground truth annotations, and inappropriate use of evaluation metrics. Through detailed empirical analysis, we uncover these issues profound impact on the evaluation and ranking of segmentation methods. Drawing from our findings, we propose a set of actionable recommendations to establish fair and robust evaluation standards for topology-aware medical image segmentation methods.